by David von Hippel and Doo Won Kang
6 April 2015
I. Introduction
In this Policy Forum David von Hippel and Doo Won Kang write ‘Given Koreans’ increasing appetite for ownership of road vehicles, there is an important role for a fuel that is carbon-free, portable, energy-dense, and compatible with existing cars and fueling systems. Ammonia (NH3) fulfills many of these requirements, as it produces no carbon dioxide when it is burned, is usable in existing vehicles with only modest engine modifications, is familiar to producers as an industrial and agricultural chemical traded worldwide, and requires only low-pressure tanks for storage, similar to liquefied petroleum gas (LPG, or “propane”).’
David von Hippel is a Nautilus Institute Senior Associate working on energy and environmental issues in Asia, as well as on analysis of the DPRK energy sector. Doo Won Kang is a student at Carnegie Mellon University’s Engineering School, Pittsburgh, PA, with a strong interest in research into the future of low-carbon fuels for the transportation sector
This paper was originally published with support from the Hanyang University’s Energy, Governance and Security (EGS) Center, available in Global Energy Monitor Vol. 3, No. 2 (February 2015).
The views expressed in this article are those of the author and do not necessarily reflect the official policy or position of the Nautilus Institute. Readers should note that Nautilus seeks a diversity of views and opinions on contentious topics in order to identify common ground.
II. POLICY FORUM BY DAVID VON HIPPEL and Doo Won Kang
Ammonia as a fuel for passenger vehicles: possible implications for greenhouse gas reduction in Korea
Among the goals of “green growth” in the Republic of Korea (ROK) are shifting to use of renewable fuels in place of fossil fuels. Renewable options—including solar space heating, biomass-fueled heating and power, and solar and wind displace coal and gas used in electricity generation (along with, possibly, more nuclear power) are available for many sectors, but fossil fuels are hardest to displace in the transportation sector. Fossil fuels offer a difficult-to-match combination of energy density and availability for vehicle use. Given Koreans’ increasing appetite for ownership of road vehicles, there is an important role for a fuel that is carbon-free, portable, energy-dense, and compatible with existing cars and fueling systems. Ammonia (NH3) fulfills many of these requirements, as it produces no carbon dioxide when it is burned, is usable in existing vehicles with only modest engine modifications, is familiar to producers as an industrial and agricultural chemical traded worldwide, and requires only low-pressure tanks for storage, similar to liquefied petroleum gas (LPG, or “propane”). The degree to which shifting to ammonia as a fuel fulfills green growth objectives, relative to other ways of reducing carbon emissions from road transport, depends in large part, however, on how ammonia is produced.[1]
A key goal of green growth policies in the ROK, as elsewhere, is to reduce emissions of the pollutants that lead to climate change. Climate change, and specifically, global warming, is caused by increasing concentrations of greenhouse gases (GHGs) in the atmosphere, especially carbon dioxide (CO2). The atmospheric build-up of GHGs is largely the result of combustion of fossil fuels and other human activities, as reaffirmed by the Intergovernmental Panel on Climate Change in September of 2013.[2]
A significant portion of CO2 emissions come from the tailpipes of cars and trucks. Practically all of current Korean vehicles run on gasoline and diesel, and as those fuels are burned, CO2 is released. The transportation sector contributed 12% of total ROK greenhouse gases (GHG) emissions in 2011. Within the transportation sector, road-transportation, including passenger cars, trucks and buses, contributed 95% of those emissions.[3]
A projection of the composition of the future passenger vehicle fleet in Korea prepared in late 2012 by the Korea Energy Economics Institute (KEEI) suggests that without aggressive application of measures to reduce road transport GHG emissions, those emissions will continue to increase, probably significantly faster than population.[4] KEEI’s projections show that although the ROK’s population will stabilize at about 52 million people in 2030, and begin to fall thereafter, the number of passenger transport vehicles will continue to increase, from about 13 million in 2010 to over 21 million by 2035, implying an increase in the number of cars per person from about 0.27 in 2010 to about 0.40 by 2035. Moreover, KEEI’s projections show very limited penetration of high-efficiency or alternative-fueled vehicles, with only about 1.7 percent of vehicles being hybrid (driven by both fossil-fueled and electric motors) by 2035, and with a scant 3,500 electric-only vehicles in the fleet by that year.
Reducing road vehicle GHG emissions can involve a number of potential “fixes”. Moving more transit from private vehicles to mass transit in other, more efficient forms of transportation including rail, subway, and buses, is one approach, and is to some extent underway in the ROK, though based on the appetite for personal transport in Korea projected by KEEI, there will be limits to the effectiveness of this “mode shifting”. How, then, can Korea achieve deep reductions in CO2 emissions from the transportation sector?
Other approaches to reducing GHG emissions by private vehicles require modifications to the makeup of the private passenger fleet itself, either through improvements in vehicle efficiency (including the dissemination of vehicles with hybrid powertrains), and the use of vehicles that use alternative fuels. One alternative fuel and powertrain combination is electric vehicles (EVs). The battery technologies required by EVs, however, though improving rapidly in storage capacity and falling in cost, still do not match the range and cost-effectiveness (from the standpoint of vehicle purchase costs) of gasoline and diesel-fueled vehicles. Other fuels that have received significant attention are compressed natural gas (CNG), which burns cleanly and can be used in most internal combustion engines, with some modifications, but requires high-pressure tanks for on-board gas storage, and hydrogen, which can be made using electricity and water (or from fossil fuels or biomass), and can be used in either internal combustion engines or in fuel cells that convert the hydrogen to electricity without combustion, and therefore work more like a battery than an a typical gasoline or diesel motor, and at an efficiency typically much higher than that of a typical auto engine. When hydrogen burns (or is converted to electricity in a fuel cell) water vapor is the main product. LPG is widely used in lightly-modified vehicles, including much of Korea’s taxi fleet, and produces slightly lower emissions than gasoline or diesel. A fifth alternative fuel is ammonia, which, like hydrogen, produces practically no GHGs when burned.[5]
Although ammonia-fueled vehicles have a number of enthusiastic proponents around the globe, most notably in the farm belt of the United States, NH3 vehicles have received generally less attention than CNG or hydrogen-fueled vehicles.[6] NH3-fueled vehicles have the potential to reduce CO2 emissions to levels far below those achieved by some alternative-fueled cars, such as those fueled with natural gas or ethanol derived from corn. The mode of operation of NH3-fueled vehicles is similar to conventional gasoline-fueled internal combustion-engine vehicles: Liquid ammonia is burned with oxygen in order to move an engine’s pistons, producing power that is harnessed to drive the vehicle’s wheels. This familiar technology means NH3-fueled vehicles can generally be built and maintained in the same way as the current vehicle fleet. NH3-fueled vehicles, however, unlike conventionally-fueled vehicles (and like hydrogen and electric vehicles), do not directly release any carbon dioxide.[7] Ammonia can be used in internal combustion engine (ICE) vehicles with minor modifications, and is environmentally friendly, as it produces only molecular nitrogen (N2) and water (H2O) at the tailpipe, even when only low-cost emissions controls are used. Any unburned ammonia and NOx in the engine’s exhaust are removed by a selective catalyst reduction (SCR) system in NH3-fueled vehicles.[8]
Recent research suggests that ammonia could also be used as a high-density, low-pressure means of storing hydrogen, with a compact on-board conversion device producing hydrogen for fuel cell vehicles with low, or possibly no, nitrogen oxide (NOx) emissions.[9]
Compared to gasoline vehicles, NH3-fueled vehicles do not produce CO2 during operation. When GHG emissions from vehicles are considered, however, it is important to look at not just the direct emissions associated with vehicle operation, but at the full energy-cycle emissions associated with fueling the vehicles. A full consideration of emissions of electric vehicles, for example, must include the emissions associated with producing and delivering the electricity stored in vehicle batteries. Similarly, a full accounting of GHG emissions from CNG vehicles must include emissions from gas production, processing, transportation, distribution, and compression. GHGs from hydrogen-fueled vehicles should include emissions associated with hydrogen production, and an accounting of GHG emissions from gasoline, diesel, and LPG vehicles should include not only emission from the tailpipe, but from oil refining and product distribution. Similarly, an accounting of GHG emissions from NH3-fueled vehicles must include the GHGs associated with NH3 manufacture. Current industrial ammonia production plants run principally on fossil fuels, most commonly natural gas and emit approximately 1.2 – 1.8 metric tons of CO2 per ton of ammonia produced.[10] Ammonia can be and is also, however, produced using electricity through the catalytic reaction of nitrogen from air (which is 78 percent N2) and hydrogen from water. Current industrial electricity-to-NH3 production is somewhat over 50 percent efficient, but once advanced ammonia production methods (such as solid state ammonia synthesis) that are now working at the lab scale are commercialized, with the use of electricity from non-fossil sources (renewable energy sources or nuclear power), virtually no CO2 emissions will be emitted during ammonia production process, with only modest emissions even including, for example, GHGs associated with power plant construction and operation. The same, of course, applies to fuel sources for electric or hydrogen-fueled vehicles.
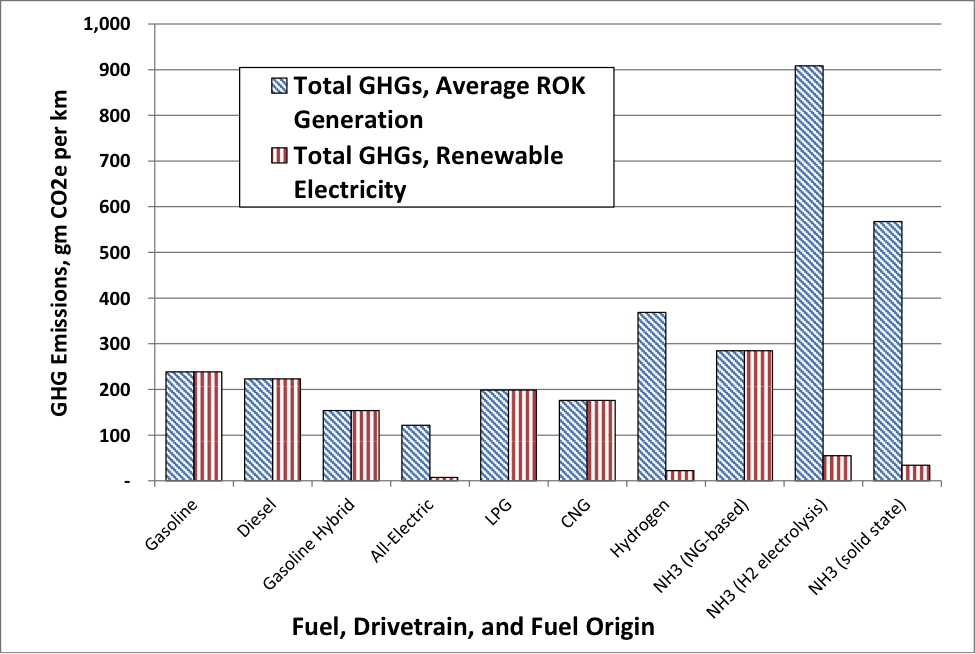
The graph below presents the authors’ estimates of the total GHG emissions per kilometer, estimated over the full energy cycle, including fuel extraction, transmission, distribution, refining, electricity generation, fuel consumption, and, for generation facilities, emissions related to fuel production and power plant construction/operations. The vehicles shown are illustrative example chosen to be generally comparable—most are commercially-available compact and, in one case, medium-sized sedans. For vehicles using electricity (“All-electric”) or fuels derived from electricity (“Hydrogen”, “NH3 (H2 electrolysis)”, and “NH3 (solid state)”), emissions were estimated in two ways, first using emission factors related to the average generation fleet in the ROK as of 2012 (blue bars),[11] and second, assuming renewable generation in a 50/50 wind/solar PV mix (red bars). Several conclusions are clear from this graph. First, electric vehicles offer the lowest emissions per km. Second, NH3 and H2 vehicles in which electricity is used to produce the fuel have higher energy-cycle emissions because of the conversion losses in electricity generation (coupled with the lower efficiency of internal combustion, relative to electric drive, in ammonia-fueled vehicles).[12] Third, in order for H2 and NH3 vehicles to be competitive with other vehicles on an overall GHGs-per-km basis, their fuels must be made using fossil-free electricity.
That said, other considerations, including cost of vehicles, costs of fuel production, vehicle range, fuel safety, [13] [14] and adaptability of fuels to existing vehicles, will also play roles as the ROK and global vehicle fleets evolve. The advantages of ammonia as a motor fuel—including its portability, compatibility with familiar fueling systems, existing industrial infrastructure, and the ability of conventional cars to easily be modified to run on a mixture of up to 85 percent ammonia,[15]—make the concept of NH3-fueled vehicles and companion NH3-from-renewable-energy production technologies well worth pursuing.
Title image: SeongJoon Cho/Bloomberg
III. References
[1] This article is based in part on Doo Won Kang (2014), “Combating climate change with ammonia-fueled vehicles”, Bulletin of the Atomic Scientists, 17 February 2014, available at http://thebulletin.org/combating-climate-change-ammonia-fueled-vehicles.
[2] Intergovernmental Panel on Climate Change, 27 September 2013. IPCC Fifth Assessment Report (WGI AR5).
[3] “2013 National Greenhouse Gas Inventory Report of Korea,” Greenhouse Gas Inventory & Research Center of Korea, Feb, 2014 (Korean). Available as http://www.gir.go.kr/home/board/read.do;jsessionid=9Y0S23c1aQqsFwoLR1oxYgiIfTaLz1jsVXTJbE1FsVNzCFcYS7z09PnC78u7cAX3.og_was_servlet_engine1?pagerOffset=0&maxPageItems=10&maxIndexPages=10&searchKey=&searchValue=&menuId=36&boardId=22&boardMasterId=2&boardCategoryId=.
[4] “Analysis of the influence of dissemination of electric vehicles on Korea’s energy supply and demand,” KEEI, Dec. 2012 (in Korean).
[5] A small amount of nitrogen oxide (NOx) emissions are produced when ammonia or hydrogen are burned. NOx has an indirect impact on GHG concentrations in the atmosphere, but the impact is much smaller than direct emissions of CO2 from fossil fuels.
[6] See, for example, the presentations prepared for the 11th Annual NH3 Fuel Conference, “NH3, the Renewable Carbon Free Fuel”, held September 21 – 24, 2014 in Des Moines, Iowa, USA, and available as http://nh3fuelassociation.org/events-conferences/2014-nh3-fuel-conference/.
[7] N. Olson and J. Holbrook, Iowa Energy Center (2012), NH3 – “The Other Hydrogen”, available from: http://www.iowaenergycenter.org/grant-and-research-library/nh3-the-other-hydrogen-report.
[8] William Jacobson, Gasoline/Ethanol/Ammonia Mixture as a Transition Fuel “Solution”, SY-Will Engineering, Available from: http://www.sy-will.spyang.com/.
[9] Autoblog (2014), “Is ammonia the secret to better hydrogen cars?”, dated June 30th , 2014 , and available as http://www.autoblog.com/2014/06/30/ammonia-secret-to-better-hydrogen-cars/.
[10] Jason C. Ganley, John H. Holbrook, Doug E. McKinley, “Solid State Ammonia Synthesis,” 2007 Annual NH3 Fuel Conference, San Francisco, CA, Oct. 15-16, 2007, available as http://www.claverton-energy.com/wordpress/wp-content/files/NHThree_SSAS_Oct2007_Final.pdf.; Sam Wood and Annette Cowie, “A Review of Greenhouse Gas Emission Factors for Fertiliser Production,” June 2004, available as http://task38.org/publications/GHG_Emission_Fertilizer_Production_July2004.pdf.
[11] Data from KEEI (2013) 2013 Yearbook of Energy Statistics, pages 172 through 177. Available as http://www.keei.re.kr/keei/download/YES2013.pdf.
[12] Note that using NH3 in a hybrid vehicle, in this comparison, would reduce emissions by roughly a third from those shown.
[13] Nijs Jan Duijm, Frank Markert, Jette Lundtang Paulsen, “Safety assessment of ammonia as a transportation fuel,” Riso National Laboratory, Denmark, February 2005; “Comparative Quantitative Risk Assessment of Motor Gasoline, LPG and Anhydrous Ammonia as an Automotive Fuel,” Quest Consultants Inc., June 2009. Available as http://www.iowaenergycenter.org/wp-content/uploads/2012/03/NH3_RiskAnalysis_final.pdf.
[14] George Thomas and George Parks (2006), Potential Roles of Ammonia in a Hydrogen Economy: A Study of Issues Related to the Use Ammonia for On-Board Vehicular Hydrogen Storage, US DOE, February 2006. Available as http://www.hydrogen.energy.gov/pdfs/nh3_paper.pdf.
[15] Helen Knight, “Portable ammonia factories could fuel clean cars,” NewScientist, 01 September 2011. Available as http://www.newscientist.com/article/mg21128285.100-portable-ammonia-factories-could-fuel-clean-cars.html#.VLrGKC7ruPU.